Anastrozole is an aromatase inhibitor used to treat hormone receptor-positive breast cancer in postmenopausal women. Anastrozole STELLA 1 mg is a film-coated tablet, 1 mg (1 tablet) taken orally once daily and should be used as directed by a doctor.
Letrozole STELLA 2.5 mg (Letrozole 2,5 mg) belongs to antineoplastic and immunomodulating agents, aromatase inhibitors, indicated in adjuvant treatment of postmenopausal women with hormone receptorpositive invasive early breast cancer.
Capecitabine STELLA 500 mg (Capecitabine 500 mg) is an oral cancer drug, indicated for the treatment of colon cancer, stomach cancer, and breast cancer, may be used alone or in combination with other drugs depending on the stage of the disease.
Ondanstella 8 (Ondansetron 8 mg) is an antiemetic drug belonging to the 5-HT3 receptor antagonist class. It works by blocking nausea signals from reaching the brain and is primarily indicated for the prevention and control of nausea caused by chemotherapy, radiation therapy, or surgery.
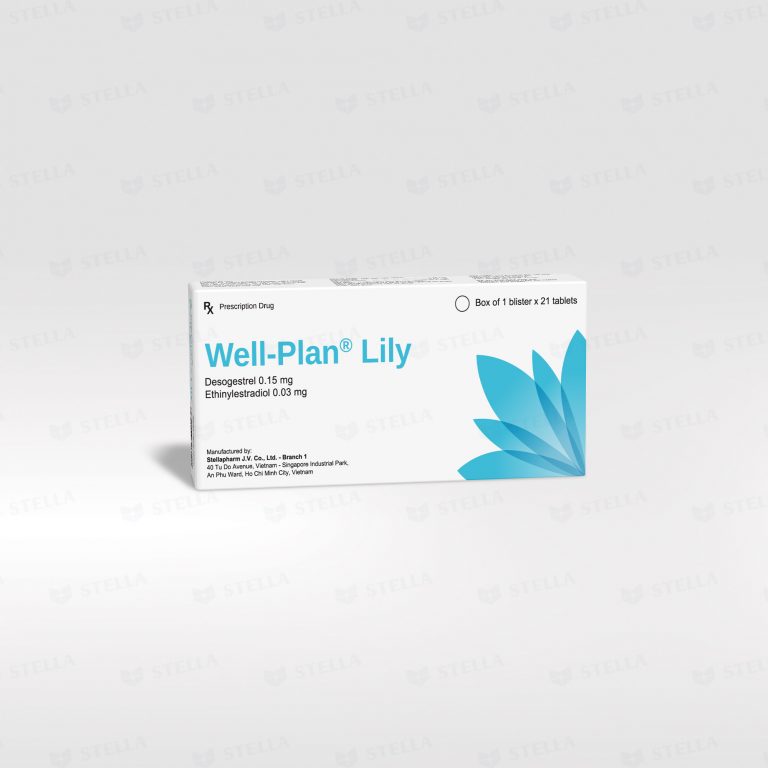
Each Well-Plan® Lily daily oral contraceptive pill contains 0.15 mg desogestrel and 0.03 mg ethinylestradiol. Before prescribing, Doctor should carefully assess risk factors, particularly the likelihood of venous blood clot formation, and compare it with other combined hormonal contraceptives.
Well-Plan® Jasmine is a combined oral contraceptive with ethinylestradiol and the progestogen drospirenone. It has no estrogenic, glucocorticoid and antiglucocorticoid activity. This gives drospirenone a pharmacological profile closely resembling the natural hormone progesterone.
Montelukast 10 mg is indicated for adults and children 15 years of age and older for the prevention of chronic asthma, relief of exercise-induced bronchoconstriction, and treatment of allergic rhinitis. This drug should be taken in the evening for the most effective asthma control.
Erdostella 300 (Erdosteine 300 mg) belongs to the mucolytic class of drugs that reduce the viscosity of mucus and phlegm, treating the symptoms of acute exacerbations in chronic bronchitis in adults.
Stebastin 20 (Ebastine 20mg) is indicated in treatment of symptoms of urticaria, severe allergic rhinitis, with or without allergic conjunctivitis.
Itraconazole 100 mg is a broad-spectrum antifungal medication that kills or inhibits the growth of many types of fungi. It is commonly prescribed to treat systematic fungal conditions when first-line systemic anti-fungal therapy is inappropriate or has proved ineffective. This may be due to underlying pathology, insensitivity of the pathogen or drug toxicity.
Allostella 3G (L-ornithine L-aspartate 3000 mg) oral granules is for the treatment of concomitant disease and sequelae due to impaired hepatic detoxification activity. Allostella 3G is dissolved in plenty of fluid (e.g. a glass of water, tea or juice) and taken with or after meals.
Calcium polystyrene sulfonate is an ion-exchange resin that is recommended for the treatment of hyperkalaemia associated with anuria or severe oliguria, in patients requiring dialysis and in patients on regular haemodialysis or on prolonged peritoneal dialysis.
Dapostella 30 (Dapoxetine 30 mg) is a prescriped to treat premature ejaculation in men aged 18-64. It works by inhibiting neurotransmitters, helping to control ejaculation and improve the duration of intercourse.
Tamsulosin 0.4 mg (Tamsulosin hydrochloride) is an alpha-1A blocker used to treat lower urinary tract symptoms caused by benign prostatic hyperplasia (BPH), such as difficulty urinating, frequent urination, and a weak urine stream. The drug works by relaxing the smooth muscles of the prostate and bladder neck.
Creswell 80 (Sotalol hydrochloride 80 mg) belongs to non-selective beta-adrenergic blockers (class II) and class III antiarrhythmics, are used to treat ventricular arrhythmias, atrial fibrillation, and atrial flutter. They work by slowing the heart rate, maintaining a normal sinus rhythm, and preventing the recurrence of arrhythmias.
In patients with essential hypertension, a single oral daily dose of cilnidipine showed a hypotensive action that maintained for 24 hours and was still evident early in the next morning.
Ramistell 1.25 (Ramipril 1.25 mg) is indicated for the treatment of high blood pressure, heart failure, and to reduce the risk of myocardial infarction/stroke. The drug works by dilating blood vessels, helping to lower blood pressure and reduce the burden on the heart.
Stadlacil 4 (Lacidipine 4 mg) is indicated for the treatment of hypertension as monotherapy or in combination with other antihypertensive agents such as beta-blockers, diuretics and ACE inhibitors.
In both healthy subjects and patients with hypercholesterolaemia, pravastatin sodium lowers the following lipid values: total cholesterol, LDL-cholesterol, apolipoprotein B, VLDL-cholesterol and riglycerides; while HDL-cholesterol and apolipoprotein A are elevated.
At the recommended dose, ivabradine helps reduce heart rate by approximately 10 beats per minute at rest and during exercise. Ivabradine STELLA 7.5 mg helps reduce the burden on the heart during activity and decreases the oxygen demand of the myocardium.
Ecoxia 30 (Etoricoxib 30 mg) is a non-steroidal anti-inflammatory drug (NSAID) that selectively inhibits COX-2, used to reduce pain and inflammation in osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis, acute pain. The usual dose is 30-60 mg once daily. It requires a prescription and long-term use should be avoided due to cardiovascular and gastrointestinal risks.
Eperistad (Eperisone hydrochloride) is effective in improving myotonic symptoms such as stiffness of shoulders, cervical pain, muscle contraction type headache, dizziness, lower back pain and rigidity of extremeties which may accompany encephalomyelopathies, cervical syndrome, periarthritis of the shoulder or lumbago.
Empagliflozin STELLA 25 mg (Empagliflozin 25 mg) is a medication used to treat type 2 diabetes and reduce the risk of cardiovascular death and heart failure. It works by increasing glucose excretion in the urine, typically at a dose of 10-25 mg/day, taken in the morning.
Vildagliptin is used in the treatment of type 2 diabetes in adults (Monotherapy for patients who are not adequately controlled by diet and exercise, and/or in combination with other antidiabetic agents).
Sitagliptin in Gliptinestad 100 is indicated for type 2 diabetes mellitus in patient has previously performed diet and exercise without achieving results.
Auxilprazol 40/1100 (Omeprazole 40 mg, Sodium bicarbonate 1100 mg) is indicated for adults in treatment of active duodenal ulcer, benign gastric ulcer, gastroesophageal reflux disease.
Alumastad contains dried aluminum hydroxide gel and magnesium hydroxide which are antacids pain reliever and indigestion discomfort by neutralizing acid, reducing the excess acidity in the stomach.
Simethicone does not affect gastric acid secretion or nutrient absorption. Ex-gas TAB 80 (Simethicone 80 mg) helps reduce bloating and abdominal distension caused by gas accumulation in the gastrointestinal tract in adults.
G.Samin Forte (Glucosamine sulfate potassium chloride, Chondroitin sulfate sodium, Black pepper dry extract) helps support the improvement of joint pain and mobility difficulties in adults.
Simarin plus with combination of silymarin (from Milk Thistle extract) with Vitamin B1, B6, B12 helps improve liver function and jaundice due to poor liver function.
Estrostad capsules contain herbal ingredients (Angelica gigas, Cynanchum wilfordii, and Phlomis umbrosa) that help manage premenopausal-related issues. This health supplement is not therapeutic claimed.
Tauginko 500 STELLA containing taurine and dried ginkgo leaf extract is for adults with cerebral circulatory insufficiency. This product is not approved therapeutic claims.
Ferolina (Zinc fumarate, spirulina powder, folic acid, vitamin B12) helps reduce the risk of zinc deficiency anemia, supports the process of blood formation in adults and children over 10 years old.
CoQ10-STELLA contains Coenzyme Q10 and black pepper extract, supports antioxidant activity and promotes cardiovascular health in adults. This product is not approved therapeutic claims.
VitK-D STELLA (Vitamin K2, vitamin D3) is for adults at risk of osteoporosis due to vitamin D deficiency and for children from 6 months of age at risk of rickets. This product is not approved therapeutic claims.
ZinDcal (Calcium, Magnesium, Zinc, Vitamin D3) supports enhanced calcium absorption, helps strengthen bones and teeth, aids in height development, reduces the risk of rickets in children, and decreases the risk of osteoporosis in adults.
Helix STELLA containing dried ivy leaf extract (Hedera helix L.) is used for adults and children above 1 in dry cough, productive cough, cough due to weather change, sore throat, bronchitis. This product is health supplement, not approved therapeutics claims.
Helix STELLA FORTE containing dried ivy leaf extract (Hedera helix L.) helps relieve cough and reduce phlegm.
Supplements folic acid for the body. Supports increasing blood formation. Helps reduce anemia due to folic acid deficiency,
Rutin-C STELLA (Rutin trihydrate combined with ascorbic acid) helps increase vascular endurance, supports antioxidant activity and promotes cardiovascular health in adults.
Aztezan (Azithromycin 500mg) is indicated for treatments respiratory tract infections (pneumonia, bronchitis), skin and soft tissue infections, middle ear infections, and sexually transmitted diseases. Azithromycin is well tolerated with a low incidence of undesirable effects. Aztezan is manufactured by Stellapharm under technology transfer of Auxilto Healthcare GmbH (Germany).
Calcium Auxilto 1000 mg (calcium 1000 mg) is used in prevention and treatment of calcium deficiency, in osteoporosis, in addition to vitamin D3 therapy for rickets and osteomalacia.
Clobetad (Clobetasol 0.05%) is a topical corticosteroid, commonly used in short term treatment of more resistant inflammatory and pruritic manifestations of steroid responsive dermatoses unresponsive to less potent corticosteroids.
Stellbutine (Trimebutine 100 mg) is indicated for the treatment of gastrointestinal dysfunction, specifically for the symptomatic relief of pain, bowel motility disturbances, and intestinal discomfort associated with gastrointestinal dysfunction.
Rebagastrin (Rebamipide 100 mg) belongs to the group of drugs used for treatment gastric and duodenal ulcers and gastroesophageal reflux disease in adults.
Racecadotril granules for oral suspension is prescribed for complementary symptomatic treatment of acute diarrhoea in infants (older than 3 months) and in children together with oral rehydration.
Almasane is a balanced mixture of two antacids and an antiflatulent-agent simethicone, belongs to the group of drugs for acid related disorders, normally used for the symptomatic relief of dyspepsia, heartburn, flatulence.
Donestad 10 (Donepezil hydrochloride 10mg) is an acetylcholinesterase inhibitor prescribed for the treatment of mild to moderate to severe dementia symptoms in Alzheimer's disease. The usual dose is one 10mg tablet per day, taken in the evening just before bedtime.
Paracetamol 500mg is for symptomatic treatment of mild to moderate pain and/or fever states. This presentation is reserved for adults and children from 27 kg (i.e. from about 8 years old).
Paracetamol is an effective alternative to aspirin as an analgesic-antipyretic agent; however, it is not a useful agent to treat inflammatory conditions. Paracetamol lowers body temperature in patients with fever but rarely lowers normal body temperature.
Paracetamol is an effective alternative to aspirin as an analgesic-antipyretic agent; however, it is not a useful agent to treat inflammatory conditions. Paracetamol lowers body temperature in patients with fever but rarely lowers normal body temperature.
Paracetamol is an effective alternative to aspirin as an analgesic-antipyretic agent; however, it is not a useful agent to treat inflammatory conditions. Paracetamol lowers body temperature in patients with fever but rarely lowers normal body temperature.
Partamol-Cafein is fixed-combination of paracetamol and caffeine that is indicated for treatment of most painful and febrile conditions, for example, headache, including migraine, backache, toothache.....
Partamol 150 Supp. (paracetamol 150 mg) is reserved for symptomatic treatment of mild to moderate pain and/or feverish states in infants weighing 8 to 12 kg (approximately 6 to 24 months).
Stadsidon 20 (ziprasidone 20 mg) is indicated in treatment of bipolar I disorder, treatment of schizophrenia and acute treatment of agitation in schizophrenia.
Stadolac 200 (etodolac 200mg) is a nonsteroidal anti-inflammatory drug (NSAID) that exhibits antiinflammatory, analgesic, and antipyretic activities. It is indicated for the management of pain in osteoarthritis, rheumatoid arthritis.
Stadlofen 100 Supp (Diclofenac sodium 100 mg) rectal suppositories are used in adults and the elderly to reduce inflammation and moderate to severe pain. The medication is effective in treating rheumatoid arthritis, osteoarthritis, ankylosing spondylitis, and acute gout attacks.
S-Profen 400 (Dexibuprofen 400 mg) is an anti-inflammatory and anti-rheumatic product, non-steroids, has an analgesic, anti-inflammatory and anti-pyretic effect in osteoarthritis.
Fencopain 50 (Flurbiprofen 50 mg) is used in the treatment of arthritis. Additionally, it helps reduce symptoms in musculoskeletal disorders and injuries. Fencopain 50 also provides relief from mild to moderate pain, including toothache, post-surgical pain, menstrual cramps, and migraines.
Uprofen 400 (Ibuprofen 400 mg) is an NSAID pain reliever and anti-inflammatory drug commonly used to reduce fever, relieve headaches, menstrual cramps, and mild to moderate musculoskeletal pain. It should be taken after a full meal to minimize stomach irritation.
Naproflam 500 (Naproxen 500 mg) is a non-steroidal anti-inflammatory analgesic compound with antipyretic properties. Naproxen inhibits prostaglandin synthetase (as do other NSAIDs).
Aldenstad plus (Alendronic acid; Cholecalciferol) is commonly used for treatment of bone diseases. It is indicated for the treatment of postmenopausal osteoporosis in women at risk of vitamin D insufficiency. It reduces the risk of vertebral and hip fractures.
Castella 90 (Ticagrelor) is co-administered with acetylsalicylic in the prevention of atherothrombotic events in adult patients with acute coronary syndromes and with history of myocardial infarction and a high risk of developing an atherothrombotic event.
Simvastatin and ezetimibe combination along with a proper diet to help lower "bad" cholesterol and fats (such as LDL-C, triglycerides) and increase "good" cholesterol (HDL-C) in the blood to help reduces the risk of heart disease, an intervention in individuals at significantly increased risk for atherosclerotic vascular disease due to hypercholesterolemia.
Amiodarone is an antiarrhythmic drug used to treat and prevent certain types of arrhythmias. These arrhythmias include ventricular tachycardia, ventricular fibrillation, and wide QRS tachycardia, as well as atrial fibrillation and paroxysmal supraventricular tachycardia. However, evidence in cardiac arrest is limited.
Nitroglycerin belongs to the group of organic nitrates. The Nitrostad 0.6 sublingual tablet containing 0.6 mg of nitroglycerin has a vasodilatory effect, helping to improve blood circulation, thereby preventing the recurrence of angina (due to inadequate blood supply to the heart)
Stefamlor 5/10 (amlodipine 5 mg, atorvastatin 10 mg) is indicated for the prevention of cardiovascular events in hypertensive patients with 3 associated cardiovascular risk factors, with normal to moderately elevated cholesterol without established coronary artery disease, and in whom, according to current recommendations, the concomitant use of amlodipine and a low dose of atorvastatin is suitable.
Rupatadine STELLA 10 mg (Rupatadine fumarate) is designed for symptomatic treatment of allergic rhinitis and urticaria in adults and adolescents (over 12 years of age).
Bilazin 20 (Bilastine) is indicated in symptomatic treatment of allergic rhino-conjunctivitis (seasonal or perennial) and urticaria. The drug does not cause drowsiness.
Amxolstad 30 mg/10 ml has mucokinetic and expectorant properties in acute and chronic diseases of the respiratory tract accompanied by abnormal bronchial secretions.
Clenmysol Sp. (Ambroxol hydrochloride, Clenbuterol hydrochloride) is a medicine for treating respiratory diseases, reducing wheezing and improving shortness of breath, and dissolving phlegm, especially in debilitated patients.
Nordrone 5 (Norethindrone 5 mg) is a synthetic progestin hormone indicated in treatments endometriosis, amenorrhoea, dysfunctional bleeding, premenstrual syndrome, cyclical mastopathy, timing of menstruation.
Dequalinium chloride is a quaternary ammonium compound with broad-spectrum antiseptic, antifungal, and antimicrobial effects, most commonly available as 10mg vaginal suppositories (e.g., Aliquina) to treat bacterial and fungal vaginal infections and Trichomonas infections. The drug acts locally, is fast-acting, has minimal systemic absorption, and is safe for pregnant and breastfeeding women.
Vastad (Metronidazole, nystatin and neomycin sulfate) belongs to antiinfectives and antiseptics pharmacotherapeutic group, excluding combinations with corticosteroids, effective treatment for vaginitis caused by all factors, including bacteria, trichomonas, and fungi.
Progestad VT 100 is indicated to support the luteal phase as part of "Assisted Reproductive Technology" (ART) – a treatment program for infertile women.
Vinpocetine is indicated in treatment of Cerebral circulatory disorders of various origins. Alleviation of the mental and neurological symptoms of cerebral circulatory disorders. Treatment of certain ear and eye diseases with vascular causes, or relieve symptoms of these diseases.
Sulpistad 100 (Amisulpride 100 mg) is indicated for the treatment of acute and chronic schizophrenic disorders, in which positive symptoms (such as delusions, hallucinations, thought disorders) and/or negative symptoms (such as blunted affect, emotional and social withdrawal) are prominent, including patients characterised by predominant negative symptoms.
Flustad 75 (Oseltamivir) is an antiviral drug for systemic use indicated for treatment and prevention of influenza in adults and children (including full term neonates) following contact with an infected person influenza but not a substitute for influenza vaccination.
Lesovir is a fixed-dose combination of ledipasvir and sofosbuvir which are direct-acting antiviral agents against the hepatitis C virus.
Molnupiravir STELLA 400 mg (Molnupiravir 400 mg) is indicated for the treatment of mild to moderate COVID-19 disease in adults with a positive diagnostic test for SARS-CoV-2 and with at least one risk factor for severe disease progression.
Molnupiravir is indicated for the treatment of mild to moderate COVID-19 disease in adults with a positive diagnostic test for SARS-CoV-2 and with at least one risk factor for severe disease progression.
Stadfovir 25 (Tenofovir alafenamide fumarate) is indicated for the treatment of chronic hepatitis B in adults and adolescents (aged 12 years and older with body weight at least 35 kg).
Tefostad T300 (Tenofovir disoproxil fumarate) is used in conjunction with other antiretroviral agents (should not be used alone) in the treatment of human immunodeficiency virus type 1 (HIV-1) infection in adults, post-exposure prophylaxis of HIV infection and in treatment of chronic hepatitis B in adults.
Bisostad 5 (Bisoprolol fumarate 5 mg) is used to treat hypertension and angina pectoris. It is also used as an adjunct to standard therapy in patients with stable chronic heart failure.
Stadovas 5 CAP (Amlodipine 5 mg) is used to treat hypertension, angina. The drug may be used as monotherapy or in combination with other antianginal medicinal products.
Nifedipin T20 retard (Nifedipine 20 mg) is used in treatment of the prophylaxis of angina, if especially element vasospastic as Prinzmetal's angina and treatment of hypertension.
Felodipine STELLA 5 mg retard (Felodipine 5 mg) is indicated for management of hypertension and prophylaxis of stable angina pectoris. It has anti-anginal and anti-ischemic effects due to improved myocardial oxygen supply/demand balance.
Statripsine oral/sublingual tablets (Alphachymotrypsine 4.2 mg) are used to treat edema after trauma or surgery (soft tissue injury, acute trauma, sprains, contusions, hematomas, bruises, infections, eyelid edema, cramps, and sports injuries). Statripsine is also used to loosen upper respiratory tract secretions in patients with asthma, bronchitis, lung diseases, and sinusitis.
Calcium STELLA 500 mg effervescent tablets with active ingredient calcium are used in the prevention and treatment of calcium deficiency, preventing osteoporosis, rickets... for adults and children.
Bicimax (Vitamin B Complex, Vitamin C, Calcium and Magnesium) is used for supplementation of deficiencies of the vitamins B complex, vitamin C, calcium and magnesium: e.g., prolonged stress, prolonged infection and fever, intestinal diseases, gastrectomy, alcohol abuse.