January 20, 2022
Life value
STELLAPHARM was born to care and protect patient’s health, to help enhancing their lives and living longer. Your health, for today and for future.

HÀ NỘI — The Drug Administration of Việt Nam under the Ministry of Health has issued the list of three COVID-19 treatment drugs produced in Việt Nam that contain the antiviral ingredient Molnupiravir, which have been granted certificates of registration for conditional use. The three products – Molravir 400mg produced by Boston Việt Nam Pharma (based in
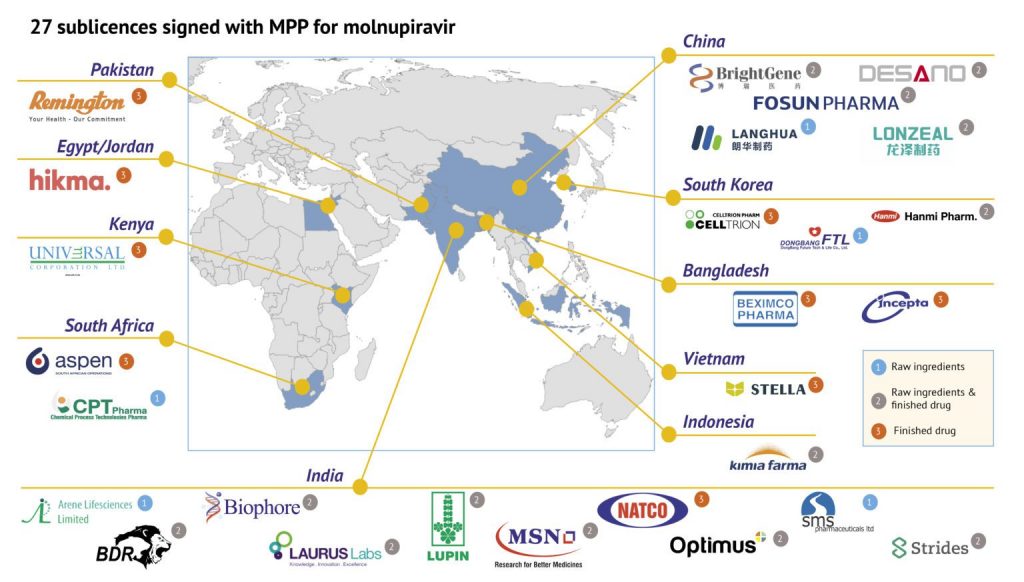
20 January 2022 Geneva – The Medicines Patent Pool (MPP) announced today that it has signed agreements with 27 generic manufacturing companies for the manufacturing of the oral COVID-19 antiviral medication molnupiravir and supply in 105 low- and-middle-income countries (LMICs). The sublicence agreements are the result of the voluntary licensing agreement signed by MPP and